Deep beneath the earth in Naica, Mexico, lies a geological marvel unlike any other: the Cave of Crystals. Discovered by miners in 2000 while excavating for ore deposits, this hidden chamber is home to colossal gypsum crystals, some of the largest natural crystals ever found. Imagine a cave filled with gleaming, milky-white crystal beams, thicker than telephone poles and reaching lengths of up to 12 meters. This is the breathtaking reality of the Crystal Cave Mexico, a subterranean spectacle that captivates scientists and nature enthusiasts alike.
Located 290 meters below a mountain rich in minerals like lead, zinc, and silver, the Crystal Cave Mexico, or Cueva de los Cristales in Spanish, has become a focal point for researchers worldwide. Drawn by its extraordinary beauty and scientific enigma, experts like Juan Manuel García-Ruiz, a crystallographer from the University of Granada, have ventured into the cave’s challenging environment to unlock the secrets of its formation and growth. For García-Ruiz, who has dedicated his life to studying crystals, witnessing the Naica crystals firsthand was a career highlight, evoking “euphoria” upon his first entry.
Over the past two decades, scientists have braved the cave’s extreme heat and humidity to unravel the mysteries surrounding these giant crystals. Now, with many questions answered, the focus has shifted towards preserving this unique geological treasure for future generations, contingent on the ongoing mining activities and natural processes within the mountain.
The Genesis of Giants: An Underground Flask
The story of the Crystal Cave Mexico began approximately 26 million years ago when magma surged upwards beneath southeastern Chihuahua, Mexico. This geological event formed the mountain near Naica and infused hot, mineral-rich waters into the mountain’s limestone caverns. It was within these mineral-laden waters that the giant crystals of Naica were conceived.
The cave environment was saturated with calcium sulfate. While calcium sulfate can crystallize into various minerals, gypsum (CaSO4·2H2O), specifically the transparent, colorless variety known as selenite, became the dominant mineral within these caves.
 Photgraph shows a person in the Cave of Crystals with giant white gypsum crystals criss-crossing behind him.
Photgraph shows a person in the Cave of Crystals with giant white gypsum crystals criss-crossing behind him.
Alt text: Exploring the Giant Selenite Crystals in Mexico’s Cave of Crystals.
Alexander Van Driessche, a crystallographer from the National Center for Scientific Research’s Institute of Earth Science, who collaborated with García-Ruiz on studying the Naica crystals, explains the mineral stability zones. Anhydrite (CaSO4) is more stable above 58 °C, while gypsum is more stable—less soluble—below this temperature.
Initially, anhydrite deposits formed in the hot, magma-heated waters. Over millennia, as the water gradually cooled below 58 °C, the anhydrite began to dissolve, and gypsum crystals started to nucleate and grow. The dissolving anhydrite provided a continuous supply of calcium and sulfate to the subterranean solution, maintaining a state of slight supersaturation—ideal conditions for the slow and steady growth of gypsum crystals ( Geology 2007, DOI: 10.1130/G23393A.1).
Whether in a laboratory or a natural cave, crystal formation always begins with nucleation. This is the process where molecular building blocks arrange themselves around a tiny nucleus and initiate growth. For instance, atmospheric particles act as nucleators for ice crystals that become snowflakes. In ionic solutions, high supersaturation leads to numerous small crystals, whereas low supersaturation favors fewer but larger crystals. The Crystal Cave Mexico offered the perfect conditions for a select few crystals to nucleate and expand to colossal dimensions over vast periods.
Van Driessche and his team’s fascination with the Naica crystals led them to conduct lab studies on gypsum nucleation. Their research revealed an unexpected nucleation process: gypsum crystals develop from coalescing nanoclusters of CaSO4, rather than from a traditional tiny gypsum crystal seed (Science 2012, DOI: 10.1126/science.1215648).
Related: Unveiling the Secrets of Giant Crystal Growth
Understanding the fundamental mechanisms of gypsum crystal formation extends beyond the caves of Mexico. It could provide insights into preventing mineral buildup in desalination plants or explain gypsum formation on Mars. However, Van Driessche and García-Ruiz were not only intrigued by nucleation but also by the remarkable size these crystals attained.
The Infinitesimal Pace of Growth
The Crystal Cave Mexico isn’t the only cave in the Naica region containing gypsum crystals; nearby chambers also hosted the same mineral-rich waters. Yet, these other caves did not produce crystals of comparable size. Van Driessche and García-Ruiz discovered that in addition to the conducive conditions for initial formation, a precisely slow cooling rate was crucial for the crystals to reach their mammoth proportions.
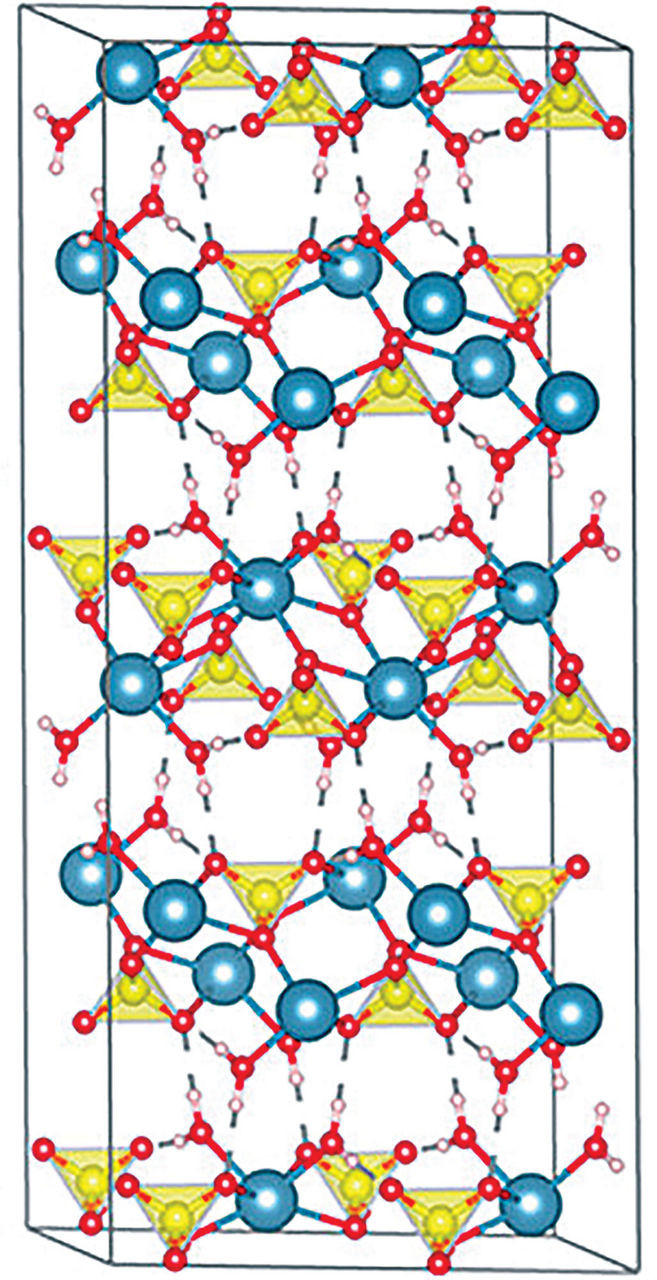 Image shows the crystal structure of gypsum.
Image shows the crystal structure of gypsum.
Alt text: Gypsum Crystal Structure: Calcium Sulfate Layers and Water Molecules.
The Cave of Swords, located at a shallower depth of 120 meters, provides a striking contrast. As its name suggests, its walls are densely covered in shorter gypsum crystals, resembling medieval swords up to 2 meters long. In the deeper Crystal Cave Mexico, the water cooled at a much slower pace compared to the Cave of Swords. This gradual cooling over eons meant that fewer gypsum crystals nucleated, allowing those few to grow to enormous sizes, according to Van Driessche. The faster cooling in the Cave of Swords resulted in a greater number of smaller crystals. He describes this as a “textbook example of crystal nucleation and crystal growth.”
Because the water temperature in the Crystal Cave Mexico remained within the narrow transition zone between anhydrite and gypsum for an extended period, the crystals experienced uninterrupted growth for hundreds of millennia, hidden from discovery. But precisely how long did this process take? Determining the age was challenging due to the crystals’ high purity, which limited the effectiveness of isotope-dating techniques. Instead, Van Driessche, García-Ruiz, and their team meticulously measured crystal growth rates in the lab using samples from the Naica mine and its water.
The layered structure of gypsum crystals—calcium sulfate layers interleaved with water molecule layers—simplified this task. The weak hydrogen bonds between water layers allowed for easy removal of flakes, providing pristine, flat surfaces for growth studies, Van Driessche explains.
“When I entered the first time, after the first couple of minutes of stupor, I burst out laughing. I was euphoric.”
Juan Manuel García-Ruiz, crystallographer, University of Granada
By placing these pristine gypsum samples in mine water for 24–48 hours, the team measured the increase in height of the flat surface using phase-shifting interferometry. This light-based technique is sensitive enough to detect surface growth rates as slow as 10–5 nm/s. By conducting measurements at various temperatures, the scientists estimated growth rates for the Naica crystals within their formation temperature range of 54 to 58 °C. From these rates, they calculated the time required for a 1-meter-thick crystal to grow at 55 °C.
The result was astonishing: approximately nearly 1 million years (Proc. Natl. Acad. Sci. U.S.A. 2011, DOI: 10.1073/pnas.1105233108). This incredibly slow growth rate is comparable to adding the thickness of a sheet of paper every 200 years, according to Van Driessche.
A Drained and Delicate Environment
Had the Crystal Cave Mexico remained submerged, the crystals might have continued to grow even larger. The current conditions, with a water temperature of around 55 °C at that depth, are still conducive to gypsum crystal formation and growth, as evidenced by newly formed crystals in small ponds within the mine, notes Van Driessche. However, to facilitate mining operations for lead, zinc, and silver, the water level in the Naica mine has been artificially lowered over the years. While this drainage made the giant crystals accessible, it also exposed them to a harsh environment and potential degradation.
 Image shows a person in the narrow Cave of Swords with swordlike crystals covering the walls on both sides.
Image shows a person in the narrow Cave of Swords with swordlike crystals covering the walls on both sides.
Alt text: Cave of Swords Mexico: Walls Covered in Shorter Gypsum Crystals.
Industrias Peñoles, the mining company, pumped water out of the mountain at a rate of roughly one Olympic-sized swimming pool every 40 minutes, creating a small artificial lake near Naica. As the water table receded, more caves, including the Crystal Cave Mexico, became accessible to air and humans. Unlike the shallower Cave of Swords, discovered in 1910 and subsequently plundered by collectors, the Crystal Cave Mexico, due to its depth and recent discovery, was largely protected from such exploitation.
Peñoles implemented strict access controls to the Crystal Cave Mexico to safeguard both the crystals and visitors. The cave’s conditions are extreme, with temperatures around 50 °C and humidity exceeding 90%. In such an environment, sweating offers no cooling relief, pushing human physiology to its limits.
“I just want to see them once more.”
María Elena Montero-Cabrera, researcher, Center for Research in Advanced Materials
Exploring the cave is also precarious, explains María Elena Montero-Cabrera, a researcher at the Center for Research in Advanced Materials in Chihuahua. Researchers had to navigate over condensation-slick gypsum spears, requiring extreme caution to avoid falls or getting lost, as rescue operations would be incredibly challenging and dangerous.
Due to these harsh conditions, researchers could only endure 10–15 minute sessions inside the cave. The cave is sealed off from the rest of the mine by double doors, maintaining a tolerable temperature and humidity in the antechamber and protecting the crystals. Medical checks were mandatory before each entry to ensure visitors were fit to withstand the cave’s climate, Montero-Cabrera adds.
Beyond the challenges for researchers, the drained environment poses a threat to the crystals themselves. The largest beams, estimated to weigh 40–50 metric tons, risk cracking under their own weight without the buoyant support of water. Gypsum’s softness (a Mohs hardness of 2) makes it vulnerable; human foot traffic has already visibly darkened pathways on the cave floor crystals, notes Van Driessche.
Related: Dehydration Threatens Giant Crystals in Mexican Cave
To determine the best preservation strategies, Montero-Cabrera and her team investigated the impact of the drained environment on the crystal surfaces. In a year-long lab experiment, they exposed Naica crystal samples to various gaseous and liquid environments to identify potential risks to their integrity and appearance.
Their findings indicated that the crystals fared slightly better in liquid environments, while gaseous environments presented a dehydration risk. They detected bassanite, a dehydrated form of calcium sulfate, on the surface of several gypsum crystals (Cryst. Growth Des. 2018, DOI: 10.1021/acs.cgd.8b00583). This suggests a gradual change in the crystals’ appearance over time. The team concluded that removing crystals from the sealed cave is not a viable preservation method, Montero-Cabrera emphasizes.
An Uncertain Future for the Crystal Cave
Research activity in the Crystal Cave Mexico has diminished as many key questions have been addressed. Mining operations closed access to the cave around 2015 due to a leak that flooded the mine faster than pumps could manage. Although water levels have risen in the mine, it’s unclear if the Crystal Cave Mexico itself has been reflooded.
 Photo shows a person inside a cave containing large, clear gypsum crystals.
Photo shows a person inside a cave containing large, clear gypsum crystals.
Alt text: Transparent Gypsum Crystals in Pulpí Geode, Spain: A Comparative Study Location.
Montero-Cabrera notes recent reports suggesting a resumption of mining activity through a different entrance, potentially reopening access to the cave for researchers. The future of the cave, whether it will flood again or remain accessible, remains uncertain. In the meantime, researchers are exploring other gypsum deposits globally, such as the gypsum crystals within an 8-meter geode in Pulpí, Spain, to further understand gypsum crystallization and growth.
Related: Gypsum Genesis: Unlocking the Secrets of Crystal Formation
García-Ruiz and Van Driessche are studying the Pulpí crystals, which are more transparent, though smaller, than those in the Crystal Cave Mexico, seeking to understand these morphological differences. García-Ruiz also aims to refine his age estimate of the Naica crystals in the future.
Despite moving on to other research, Montero-Cabrera expresses a strong desire to revisit the cave if it reopens. “I just want to see them once more,” she says, echoing the sentiment of many who have been captivated by this natural wonder.
For now, the giant crystals remain isolated—a hidden, otherworldly spectacle awaiting an unknown fate, a testament to the slow, powerful forces of nature and a fragile beauty that scientists hope to understand and preserve.